Enormous passivation effects of a surrounding zeolitic framework on Pt clusters for the catalytic dehydrogenation of propane†
Abstract
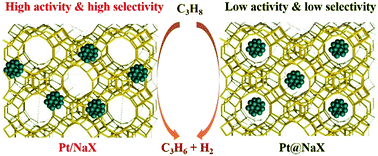
Enclosing metal clusters in zeolites can enhance their catalytic performance due to the complex effects of the zeolitic environment. However, for the first time, herein, we report the significant passivation effects of the surrounding zeolitic framework on the d-band center and catalytic performance of enclosed Pt clusters. For the dehydrogenation of propane to propylene, the Pt/NaX catalyst with Pt clusters on the exterior of X-zeolite showed a ∼1100% enhancement in TOF and largely improved selectivity for propylene compared with the corresponding Pt@NaX catalyst, in which the Pt clusters were stuck to the inside of X-zeolite. Moreover, less coke deposition and high stability were detected in Pt/NaX after the reaction for 18 h. Combining the detailed experiments and DFT calculations, it was revealed that the significant passivation effect is mainly attributed to the surrounding zeolitic framework for the enclosed Pt clusters, which decreases the d-band center of the Pt clusters and weakens the activation of propane, preventing the desorption of propylene with a reduction in propylene selectivity and increase in coke deposition. Thus, the present investigation reveals that enclosing metal clusters in zeolites may not be beneficial for some specific catalytic reactions, deepening the understanding for this type of catalyst of metal clusters enclosed in zeolites.


 Please wait while we load your content...
Please wait while we load your content...