Effect of ultra-low amount of gold in oxide-supported bimetallic Au–Fe and Au–Cu catalysts on liquid-phase aerobic glycerol oxidation in water†
Abstract
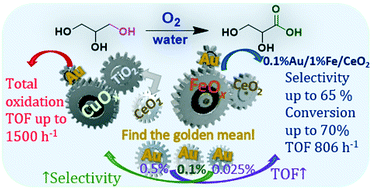
Low-loaded Au–Fe and Au–Cu bimetallic catalysts with an Au content of 0.025–0.5 wt% supported on TiO2 and CeO2 were prepared. The obtained catalytic systems revealed a unique high activity in glycerol oxidation to carboxylic acids. The results showed a strong synergetic effect between gold and base metals, which influenced the activity and selectivity of the samples. The TOF values for bimetallic catalysts exceeded 1500 h−1, and these values were much higher than the TOFs of the monometallic Au samples. Moreover, the highest oxidation activity was obtained for the systems with an ultra-low gold content of 0.025 wt%. Fe-containing bimetallic catalysts were able to provide milder oxidation of glycerol, while the Au/CuOx combination mostly led to glycerol total oxidation. The amount of gold deposited and the nature of the support used also governed the selectivity of the catalysts in glycerol oxidation to carboxylic acids. The system 0.1%Au/1%FeOx/CeO2 appeared to be the most selective to glyceric acid. The activity and selectivity of this catalyst are comparable to or even exceed the performance of noble-metal-based catalytic systems.


 Please wait while we load your content...
Please wait while we load your content...