Design of CeO2-supported LaNiO3 perovskites as precursors of highly active catalysts for CO2 methanation†
Abstract
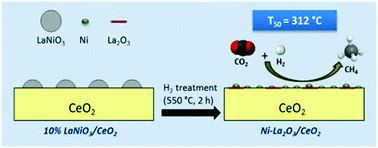
This work investigates the viability of 10–50% LaNiO3/CeO2 formulations, prepared by combined citric acid and impregnation methods, as precursors of highly active and stable materials for CO2 methanation. The prepared materials were widely characterized before and after the controlled reduction process. XRD and STEM-EDS mapping analysis confirmed the ex-solution of Ni NPs during reduction of LaNiO3/CeO2 formulations, leading to Ni–La2O3/CeO2 formation. Low LaNiO3 loading favored the ex-solution of small-sized Ni NPs (<5 nm) highly dispersed over CeO2 and La2O3 surfaces. H2-TPR experiments revealed that the higher reducibility of the samples prepared with low LaNiO3 loading promoted the H2 activation at lower temperatures. XPS experiments suggest that this promotion is due to the higher accessibility of Ni as well as Ni–ceria interaction. The material obtained after the reduction of the 10% LaNiO3/CeO2 formulation shows a higher concentration of weak–medium basic sites due to a higher accessibility of Ni NPs, La2O3 phase and Ni–CeO2 interface. The easier hydrogenation of CO2 adsorbed on these basic sites, together with the promoted H2-activation, maximized the CO2 methanation in the kinetically controlled region for this catalyst up to 71%. The intensification of Ni, La2O3 and CeO2 interactions also enhanced the CO2 methanation efficiency and the stability of the conventional 8.5% Ni/CeO2 catalyst. Thus, the 10% LaNiO3/CeO2 precursor emerges as a novel formulation to obtain highly active, selective and stable catalysts for CO2 methanation.


 Please wait while we load your content...
Please wait while we load your content...