Tuning the degradation activity and pathways of chlorinated organic pollutants over CeO2 catalyst with acid sites: synergistic effect of Lewis and Brønsted acid sites†
Abstract
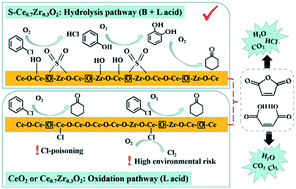
Enhancing the surface acidity has been seen as an effective strategy to optimize the performance of catalysts for the degradation of chlorinated volatile organic compounds (CVOCs). Herein, a series of catalysts are constructed by introducing Lewis and Brønsted acid sites into the skeleton surface of three-dimensional ordered macroporous (3DOM) CeO2. By a systematic study, we find that the introduction of Lewis acid sites can enhance the redox capacity of CeO2, while the surface Brønsted acid sites of the material promote chlorobenzene (CB) degradation through hydrolysis that is an effective degradation route to generate less chlorinated organic by-products. The optimal S–Ce0.7Zr0.3O2 with both abundant surface Lewis and Brønsted acid sites presents a stable activity and only 1.2% Cl2 selectivity with ∼90% CB conversion at 410 °C for 24 h. This work reveals the different roles and synergistic effects of Lewis and Brønsted acid sites to develop environmentally friendly CVOC combustion catalysts.


 Please wait while we load your content...
Please wait while we load your content...