Carbon supported Pt–Ni octahedral electrocatalysts as a model to monitor nickel corrosion and particle detachment†
Abstract
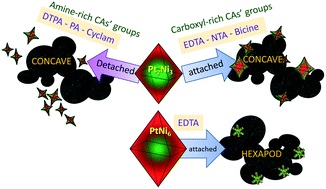
Pt–Ni nanoparticles (NPs) are used as electrocatalysts toward the oxygen reduction reaction due to their high mass activity. However, degradation processes at high potential under the acidic conditions of proton exchange membrane fuel cells delay their massive implementation. The model presented here facilitates understanding of the corrosion by clearing variables related to the electrochemical measurement (non-equilibrium conditions and complex system) and focusing on the gradual etching of Ni via two-phase Ni(II) transfer treatment promoted by chelating agents (CAs). We synthesized Pt–Ni/C octahedral NPs and then used six chemically different CAs as corrosion agents for Ni. The corrosion yielded concave octahedral and hexapod NPs. Amine-rich CA treatment results in particle detachment from the carbon substrates, while carboxylic-rich CA treatment preserves the carbon–particle bond. We correlate the detachment to the type of chemical group in the CAs and the Ni etching is correlated to the pH. The ORR of the best catalyst is reported.


 Please wait while we load your content...
Please wait while we load your content...