Bimetallic CuFe nanoparticles as active and stable catalysts for chemoselective hydrogenation of biomass-derived platform molecules†
Abstract
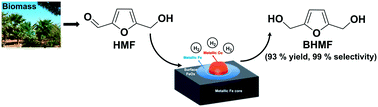
Bimetallic CuFe nanoparticles covered by thin carbon layers were developed as a potential substitute for noble metal catalysts and have been used for chemoselective hydrogenation of 5-(hydroxymethyl)furfural (HMF) to 2,5-bis(hydroxymethyl)furan (BHMF). Compared to Cu catalysts supported on conventional solid carriers prepared by impregnation, the CuFe@C nanoparticles are active and more stable catalysts. The spatial distribution of the immiscible Cu and Fe in the bimetallic CuFe@C nanoparticles is dependent on the Cu/Fe ratio, and moreover, an optimized Cu/Fe ratio has been found for hydrogenation of HMF. In the fresh Fe@C and CuFe@C catalysts, their surfaces are passivated and covered by FeOx, due to oxidation by air. Based on detailed structural characterization and catalytic studies, small Cu nanoparticles supported on Fe nanoparticles are proposed to be the key active sites for hydrogenation of HMF. These Cu nanoparticles can not only serve as the active sites for hydrogenation of HMF but also promote the reduction of FeOx into metallic Fe, resulting in an increased number of active sites in the bimetallic CuFe@C catalyst compared to the monometallic Cu@C and Fe@C samples, resulting in significant promotion of the catalytic activity.


 Please wait while we load your content...
Please wait while we load your content...