Self-sufficient asymmetric reduction of β-ketoesters catalysed by a novel and robust thermophilic alcohol dehydrogenase co-immobilised with NADH†
Abstract
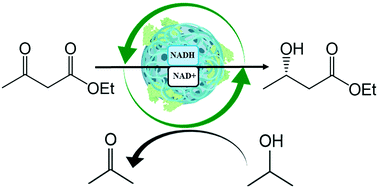
β-Hydroxyesters are essential building blocks utilised by the pharmaceutical and food industries in the synthesis of functional products. Beyond the conventional production methods based on chemical catalysis or whole-cell synthesis, the asymmetric reduction of β-ketoesters with cell-free enzymes is gaining relevance. To this end, a novel thermophilic (S)-3-hydroxybutyryl-CoA dehydrogenase from Thermus thermophilus HB27 (Tt27-HBDH) has been expressed, purified and biochemically characterised, determining its substrate specificity towards β-ketoesters and its dependence on NADH as a cofactor. The immobilization of Tt27-HBDH on agarose macroporous beads and its subsequent coating with polyethyleneimine has been found the best strategy to increase the stability and workability of the heterogeneous biocatalyst. Furthermore, we have embedded NADH in the cationic layer attached to the porous surface of the carrier. Since Tt27-HBDH catalyses cofactor recycling through 2-propanol oxidation, we achieve a self-sufficient heterogeneous biocatalyst where NADH is available for the immobilised enzymes but its lixiviation to the reaction bulk is avoided. Taking advantage of the autofluorescence of NADH, we demonstrate the activity of the enzyme towards the immobilised cofactor through single-particle analysis. Finally, we tested the operational stability in the asymmetric reduction of β-ketoesters in batch, succeeding in the reuse of both the enzyme and the co-immobilised cofactor up to 10 reaction cycles.


 Please wait while we load your content...
Please wait while we load your content...