The intrinsic effect of co-feeding water on the formation of active/deactivating species in the methanol-to-hydrocarbons reaction on ZSM-5 zeolite†
Abstract
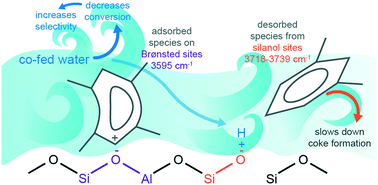
Water is formed and added in the conversion of methanol-to-hydrocarbons, slowing down both the reaction and deactivation rates. This work aims to clarify the selective nature of water quenching on a ZSM-5 zeolite catalyst in terms of (1) reaction/deactivation using an integral reactor (full range of conversions) and (2) rate of formation/growth of deactivating species using two FTIR and UV-vis in situ differential reactors (conversions lower than 0.15). Our approach assesses the effect of water under comparable conversion conditions while characterizing in detail the products and intermediates of the reaction (by online and in situ analysis, and extraction measurements). The results obtained prove, in an unbiased way, that water quenches more selectively the deactivation than the reaction with moderate amounts of added water (water/methanol = 0.11 g g−1). On the other hand, in situ FTIR spectroscopy evidences that co-feeding water sweeps the retained species from the silanol sites and favors the formation of olefins as retained species, while in situ UV-vis spectroscopy proves that the rate of formation/growth of discrete retained species drop by the addition of water and the degree of this decline is severer for coke than for coke precursors or active species.


 Please wait while we load your content...
Please wait while we load your content...