Surface phase structures responsible for the activity and deactivation of the fcc MoC (111)-Mo surface in steam reforming: a systematic kinetic and thermodynamic investigation†
Abstract
The surface phase structure evolution on a fcc MoC (111)-Mo terminated surface under a H2O/H2-rich environment typical in steam reforming (SR) reactions was systematically investigated by periodic density functional theory (DFT) calculations and an ab initio thermodynamic method. The configurations of surface adsorbates (H2O*, OH*, O*, 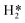 and H*) at different coverages (1/9 ML ≤ θ ≤ 1 ML) were explored. For θH2O ≤2/3 ML, the adsorption of H2O is mainly through a Mo–O coordinating interaction, while further captured H2O interacts only through hydrogen bonds with the surface coordinating H2O* at θH2O >2/3 ML. For θOH ≤4/9 ML, the surface OH* all possess cavity site configurations (coordinating with three surface Mo atoms) with relatively strong surface binding strength and significant steric hindrance, while the top site OH* with much weaker surface binding strength and small steric hindrance emerge at θOH ≥5/9 ML. For all θO, the surface O* always have cavity site configurations with obviously stronger surface binding strength than the top site OH*. The order of surface binding strength of stable adsorbates is O* (cavity site) > OH* (cavity site) > OH* (bridge site) > OH* (top site) > H2O* > H* >
and H*) at different coverages (1/9 ML ≤ θ ≤ 1 ML) were explored. For θH2O ≤2/3 ML, the adsorption of H2O is mainly through a Mo–O coordinating interaction, while further captured H2O interacts only through hydrogen bonds with the surface coordinating H2O* at θH2O >2/3 ML. For θOH ≤4/9 ML, the surface OH* all possess cavity site configurations (coordinating with three surface Mo atoms) with relatively strong surface binding strength and significant steric hindrance, while the top site OH* with much weaker surface binding strength and small steric hindrance emerge at θOH ≥5/9 ML. For all θO, the surface O* always have cavity site configurations with obviously stronger surface binding strength than the top site OH*. The order of surface binding strength of stable adsorbates is O* (cavity site) > OH* (cavity site) > OH* (bridge site) > OH* (top site) > H2O* > H* > 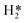 , indicating the top site OH* with moderate surface binding strength as the probable reactive surface species in SR reactions. The energy barriers and elementary reaction rates at different coverages and temperatures show that the H2O dissociation resulting in OH* is really facile, and the reaction rates are several orders of magnitude larger than those of surface O* formation by OH* dissociation, signifying that the (111)-Mo surface is more prone to be covered with OH* instead of O* at least in the initial SR reaction stage. The ab initio thermodynamic calculations under constrained-equilibrium conditions further confirm the existence of top site OH* under typical SR reaction conditions, which can deliver a reactive surface phase structure. Though the O* formation is kinetically unfavourable, its gradual accumulation by thermodynamic driving force will lead to surface Mo–O layers, the formation of which is detrimental to activity in long-term running.
, indicating the top site OH* with moderate surface binding strength as the probable reactive surface species in SR reactions. The energy barriers and elementary reaction rates at different coverages and temperatures show that the H2O dissociation resulting in OH* is really facile, and the reaction rates are several orders of magnitude larger than those of surface O* formation by OH* dissociation, signifying that the (111)-Mo surface is more prone to be covered with OH* instead of O* at least in the initial SR reaction stage. The ab initio thermodynamic calculations under constrained-equilibrium conditions further confirm the existence of top site OH* under typical SR reaction conditions, which can deliver a reactive surface phase structure. Though the O* formation is kinetically unfavourable, its gradual accumulation by thermodynamic driving force will lead to surface Mo–O layers, the formation of which is detrimental to activity in long-term running.



 Please wait while we load your content...
Please wait while we load your content...