First-principles study of a 2-dimensional C-silicyne monolayer as a promising anode in Na/K ion secondary batteries†
Abstract
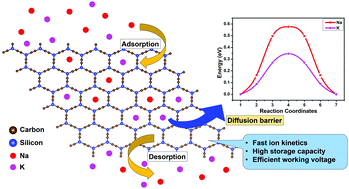
With the depleting resources of energy and increasing demand, the need for sustainable and renewable energy resources has become the need of the hour. The low storage capacity of current materials for Na/K ion batteries has led to the quest to identify suitable materials for an electrode with excellent electrochemical properties. In the present work, a systematic theoretical investigation of C-silicyne, a planar 2-dimensional hexagonal lattice, is performed to establish the geometric and thermal properties and stability. The electronic properties illustrate the metallic nature of C-silicyne, which is conserved even after the effective adsorption of Na/K ions on the surface of the monolayer. For the practical functionality, the storage capacity of C-silicyne is evaluated as 591 mA h g−1 for Na ions and 443 mA h g−1 for K ions. Moreover, the low diffusion barriers for the Na (0.57 eV) and K (0.34 eV) ions display their feasible movement across the monolayer as the electrochemical cycle progresses. The average working voltage is found to lie in the range of 0.1–1 V, which is required for the effective functioning of the anode in a Na/K ion battery. These results demonstrate the potential of C-silicyne as a material for the anode in Na/K ion batteries.


 Please wait while we load your content...
Please wait while we load your content...