Structure optimization of lipopeptide assemblies for aldol reactions in an aqueous medium†
Abstract
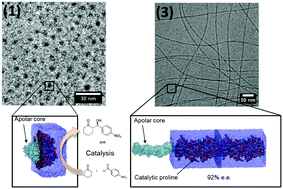
Four amphiphilic peptides were synthesized, characterized, and evaluated regarding their efficiency in the catalysis of direct aldol reactions in water. The lipopeptides differ by having a double lipid chain and a guanidinium pyrrole group functionalizing one Lys side chain. All the samples are composed of the amino acids L-proline (P), L-arginine (R), or L-lysine (K) functionalized with the cationic guanidiniocarbonyl pyrrole unit (GCP), L-tryptophan (W), and L-glycine (G), covalently linked to one or two long aliphatic chains, leading to surfactant-like designs with controlled proline protonation state and different stereoselectivity. Critical aggregation concentrations (cac) were higher in the presence of the GCP group, suggesting that self-assembly depends on charge distribution along the peptide backbone. Cryogenic Transmission Electron Microscopy (Cryo-TEM) and Small Angle X-ray Scattering (SAXS) showed a rich polymorphism including spherical, cylindrical, and bilayer structures. Molecular dynamics simulations performed to assess the lipopeptide polymorphs revealed an excellent agreement with core–shell arrangements derived from SAXS data and provided an atomistic view of the changes incurred by modifying head groups and lipid chains. The resulting nanostructures behaved as excellent catalysts for aldol condensation reactions, in which superior conversions (>99%), high diastereoselectivities (ds = 94 : 6), and enantioselectivities (ee = 92%) were obtained. Our findings contribute to elucidate the effect of nanoscale organization of lipopeptide assemblies in the catalysis of aldol reactions in an aqueous environment.


 Please wait while we load your content...
Please wait while we load your content...