On the association of frustrated Lewis pairs in ionic liquids: a molecular dynamics simulation study†
Abstract
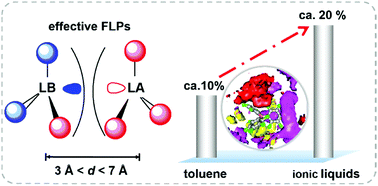
Sterically hindered frustrated Lewis pairs (FLPs) have the ability to activate hydrogen molecules, and their reactivity is strongly determined by the geometric parameters of the Lewis acids and bases. A recent experimental study showed that ionic liquids (ILs) could largely improve the effective configuration of FLPs. However, the detailed mechanistic profile is still unclear. Herein, we performed molecular dynamics (MD) simulations to reveal the effects of ILs on the structures of FLPs, in particular, the association of Lewis acids and bases. For this purpose, mixed systems were adopted consisting of the ILs [Cnmim][NTf2] (n = 6, 10, 14), [C6mim][PF6] and [C6mim][CTf3] and the typical FLP (tBu)3P/B(C6F5)3 for MD simulations. Radial distribution functions (RDFs) results show that toluene competes with (tBu)3P to interact with B(C6F5)3, resulting in a relatively low effective (tBu)3P/B(C6F5)3 complex, while [C10mim][NTf2] shows less competition with (tBu)3P, which increases the amount of effective FLPs. Spatial distribution functions (SDFs) results show that toluene forms a continuum solvation-shell, which hinders the interactions between (tBu)3P and B(C6F5)3, while [C10mim][NTf2] leaves relatively large empty spaces, which are accessible for (tBu)3P or B(C6F5)3 molecules, resulting in higher probabilities of effective FLP structures. Lastly, we find that the longer alkyl chain length of [Cnmim]+ cations, the higher the amount of effective (tBu)3P/B(C6F5)3 pairs, and the anion [CTf3]− shows negative effects, for which even less effective (tBu)3P/B(C6F5)3 pairs have been found compared to those of toluene.


 Please wait while we load your content...
Please wait while we load your content...