The role of the shell in core–shell-structured La-doped NaTaO3 photocatalysts†
Abstract
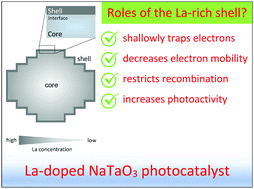
NaTaO3, a semiconductor with a perovskite structure, has long been known as a highly active photocatalyst for overall water splitting when appropriately doped with La cations. A profound understanding of the surface feature and why and how it may control the water splitting activity is critical because redox reactions take place at the surface. One surface feature characteristic of La-doped NaTaO3 is a La-rich layer (shell) capping La-poor bulk (core). In this study, we investigate the role of the shell in core–shell-structured La-doped NaTaO3 through systematic chemical etching with an aqueous HF solution. We find that the La-rich shell plays a role in electron–hole recombination, electron mobility and water splitting activity. The shallow electron traps populating the La-rich shell trap the photoexcited electrons, decreasing their mobility. The shallowly trapped electrons remain reactive and are readily available on the surface to be extracted by the cocatalysts for the reduction reaction evolving H2. The presently employed chemical etching method also confirms the presence of a La concentration gradient in the core that regulates the steady-state electron population and water splitting activity. Here, we successfully reveal the nanoarchitecture–photoactivity relationship of core–shell-structured La-doped NaTaO3 that thereby allows tuning of the surface features and spatial distribution of dopants to increase the concentration of photoexcited electrons and therefore the water splitting activity. By recognizing the key factors that control the photocatalytic properties of a highly active catalyst, we can then devise proper strategies to design new photocatalyst materials with breakthrough performances.


 Please wait while we load your content...
Please wait while we load your content...