Effect of counter-anions on the aggregation of Thioflavin-T†
Abstract
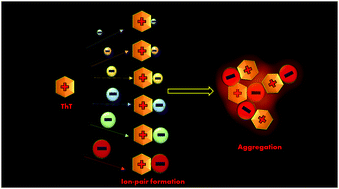
The aggregation of small molecules in aqueous solution is known to be influenced by the ionic strength of the medium; however, the role played by the identity of salt in the phenomenon of small molecule aggregation is rarely investigated. In the present contribution, we have investigated the effect of counter-anions on the aggregation of a popular cationic amyloid sensing probe, Thioflavin-T (ThT), by taking six different anions, viz. chloride, bromide, acetate, iodide, tetrafluoroborate, and perchlorate. Our results clearly indicate that it is not the ionic strength of the medium which solely controls aggregation of small molecules but distinct ions behave distinctly with regard to the organization. In fact, distinct ion effects play a major role in the salt induced organization of fluorophores. Using detailed steady-state emission, time-resolved emission, and ground-state absorption measurements, the optical properties of salt induced aggregates of ThT have been characterized. We have rationalized our observations on the basis of the theory of matching water affinity, which suggests that the matching free hydration energy is a critical aspect for the formation of contact ion pairs, which eventually results in aggregation. In brief, a larger sized anion, perchlorate, has a lower free energy of hydration and forms a suitable contact ion pair, with a larger organic cation, ThT, having weaker hydration. This contact ion-pair formation subsequently leads to the formation of an aggregate assembly which is found to be emissive in nature. Therefore, it is possible to induce aggregation of ThT by selecting the right counterion with the appropriate size, which may help us to evaluate the false positive signals when high ionic strength and specific counterions are present in the sensing matrix.
- This article is part of the themed collection: PCCP Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...