Molecular-scale origins of solution nanostructure and excess thermodynamic properties in a water/amphiphile mixture†
Abstract
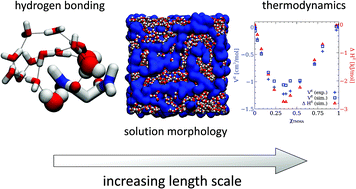
The molecular and nanoscale origins of nonideality in excess thermodynamic properties are essential to understanding cosolvent mixtures, yet they remain challenging to determine. Here, we consider a binary mixture of water and an amphiphile, N,N,N′,N′-tetramethylmalonamide (TMMA), which is characterized by strong hydrogen bonding between the two components and no hydrogen bonding between amphiphiles. Using molecular dynamics simulation, validated with excess volume measurements and X-ray scattering, we identify three distinct solution regimes across the composition range of the binary mixture and find that the transition between two of these regimes, marked by the water percolation threshold, is closely correlated with minima in the excess volume and excess enthalpy. Structural analysis of the simulations reveals an interplay between local interactions and solution nanostructure, determined by the relative strength of the water–water and water–amphiphile hydrogen bonding interactions. By comparison with other amphiphiles, such as linear alcohols, the relative strength of like and unlike interactions between water and amphiphile affects the relationship between thermodynamics and structural regimes. This provides insight into how molecular forces of mutual solvation interact across length scales and how they manifest in excess thermodynamic properties.


 Please wait while we load your content...
Please wait while we load your content...