Free energy profile analysis to identify factors activating the aggregation-induced emission of a cyanostilbene derivative
Abstract
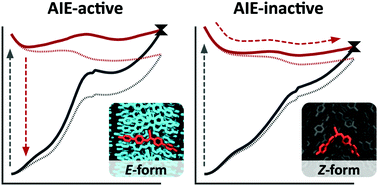
An approach to quantitatively analyze the factors contributing to the activation of aggregation-induced emission (AIE) of a molecule is proposed using molecular simulations. A cyanostilbene derivative, 1-cyano-1,2-bis-(4′-methylbiphenyl)ethylene (CN-MBE), has two isomers, E and Z forms. The E-form of CN-MBE exhibits AIE, and is non-emissive in dilute solutions but becomes highly emissive in aggregated states. The Z-form is non-emissive, even in the solid state, that is, the E-form of CN-MBE is AIE-active, while its Z-form is AIE-inactive. In this study, the free energy profiles of the AIE processes of the E and Z forms of CN-MBE are investigated using the free energy perturbation method at the quantum mechanics/molecular mechanics level. The free energy profiles reveal significant differences in the extent to which steric hindrance from surrounding molecules restricts the intramolecular motions of the E and Z forms in the aggregated states. The structural features of the E and Z forms are characterized based on the conformational changes in the excited state relaxation process to reach the conical intersections and the free volume space around the molecules in the aggregated states. This study determines the contributing factors that cause the AIE activity of the molecule by identifying characteristic differences in the free energy profiles of the AIE processes of the AIE-active E-form of CN-MBE and the inactive Z-form. The approach used in this study can be applied to the rational design of highly efficient AIE luminogens utilizing computer modeling.


 Please wait while we load your content...
Please wait while we load your content...