Entropic stabilization plays a key role in the non-uniform distribution of oxygen ions and vacancy defects in gadolinium-doped ceria†
Abstract
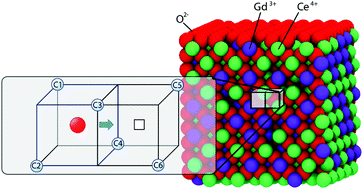
Gadolinium-doped ceria (GDC) is an important fast oxygen ion conductor. O2−-ion hopping in different cation environments of GDC is probed using molecular dynamics (MD). We find that at equilibrium, the O2−-ions and vacancy defects arrange themselves around the relatively immobile cations in a way such that pairs of O2−-ions and vacancy defect (O2−–vac pair) are in greater numbers in some cation environments than others. The difference in O2−–vac pair numbers is shown to originate from entropic effects in the cation environments. As a consequence of the entropic effect, the O2−–vac pair distribution is practically independent of temperature between 974 and 1874 K. Even the O2− ion hopping rate and barrier are influenced by the entropic effect. This observation is in stark contrast with the standard belief that energetic interactions dominate in solid oxide materials, and entropic factors can be ignored. By analyzing the cation environment effect using k-means clustering, nine environment clusters are identified that are associated with unique values of the activation barrier and entropy. Comparisons to yttria stabilized zirconia (YSZ), yet another popular fast oxygen ion conductor, are made.


 Please wait while we load your content...
Please wait while we load your content...