Sublimation – a green route to new solid-state forms
Abstract
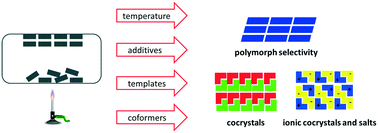
Different crystal forms of molecular solids have different physical, mechanical and chemical properties and the control of the solid-state form or the preparation of new forms (polymorphs, solvates, salts, cocrystals) is an important and challenging task in many fields ranging from pharmaceuticals, pigments, agrochemicals, and explosives to molecular electronics. The shelf-life, bioavailability and processability of an active pharmaceutical ingredient depends on the crystal form and screening for new solid-state forms including polymorphs, salts and cocrystals is now a routine part of the drug development process. A range of techniques are used to obtain different solid-state forms with high phase purity. Besides the traditional solution crystallization method these include solid-state grinding, extrusion, spray-drying and supercritical fluid methods. Recently, sublimation has emerged as a method that can give excellent crystal form selectivity and often allows the crystallization of solid-state forms that are inaccessible by other methods. In addition, sublimation is a ‘green chemistry’ technique as it produces no waste and does not use solvent. In this highlight article we summarize and discuss the literature on the crystallization of polymorphs, cocrystals and salts from the gas phase showing the tremendous potential of sublimation in solid-state form research.
- This article is part of the themed collections: 2021 Highlight article collection and Crystal Growth


 Please wait while we load your content...
Please wait while we load your content...