2,2′-Bipyridyl-1,1′-dioxide based bismuth(iii) bromide hybrids: studies on crystal structure and luminescence†
Abstract
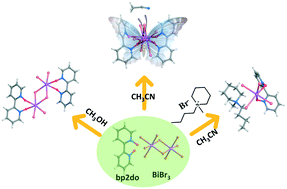
Three bismuth(III) bromide hybrids based on 2,2′-bipyridyl-1,1′-dioxide (bp2do) were obtained by a solvothermal method, namely Bi2Br6(bp2do)2 (1), Bi2Br6(bp2do)2·0.5CH3CN (2), and [PP14][BiBr4(bp2do)] (3, PP14 = 1-butyl-1-methylpiperidinium). 1 features a centrosymmetric [Bi2Br6O4] dimer, in which two identical [BiBr4O2] octahedra edge-share two Br atoms; 2 contains a [Bi2Br6O4] dinuclear unit with a unique configuration, in which a [BiBr4O2] octahedron face-shares one Br and two O atoms with a [BiBr3O4] pentagonal-bipyramid. Moreover, the metal–ligand linking mode adopted in 2 has not been reported before. The choice of organic solvents could induce different Bi–Br–bp2do geometries with a dimer in 1 and a dinuclear unit in 2. Introduction of ionic liquid cations resulted in the ionic compound 3 featuring a mononuclear [BiBr4(bp2do)] unit. A more rigid environment with more hydrogen bonds leads to visible green phosphorescence with long lifetime for 3 under UV light. The three compounds possess similar phosphorescence peaks at 77 K, which originates from inorganic-to-organic charge transfer (IOCT) as confirmed by density functional theory (DFT) calculations.
- This article is part of the themed collection: Crystal Growth


 Please wait while we load your content...
Please wait while we load your content...