Selective synthesis of enol ethers via nickel-catalyzed cross coupling of α-oxy-vinylsulfones with alkylzinc reagents†
Abstract
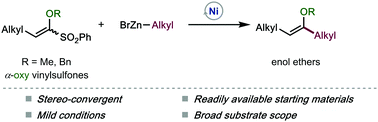
We describe here a Ni-catalyzed Negishi coupling reaction to prepare 1,2-dialkyl enol ethers in a stereoconvergent fashion. This method employs readily available and bench-stable α-oxy-vinylsulfones as electrophiles. The C–sulfone bond in the α-oxy-vinylsulfone motif is cleaved chemoselectively in these reactions. The mild conditions are tolerant of a variety of functional groups on both partners, thus representing a general strategy for enol ether synthesis. This unique reactivity of α-oxy-vinylsulfones indicates their further application as electrophilic partners in cross-coupling reactions.
- This article is part of the themed collection: 2021 Emerging Investigators


 Please wait while we load your content...
Please wait while we load your content...