Iridium-catalyzed enantioselective addition of an N-methyl C–H bond to α-trifluoromethylstyrenes via C–H activation†
Abstract
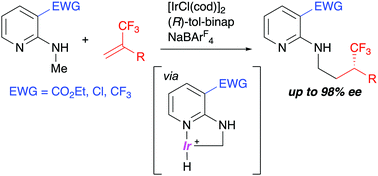
The Ir-catalyzed enantioselective addition of an N-methyl C–H bond of 2-(methylamino)pyridine derivatives to α-trifluoromethylstyrenes proceeded via C–H activation to give chiral γ-branched amine derivatives having a trifluoromethyl-substituted stereocenter. It was found that a bulky and electron-withdrawing group at the 3-position of 2-(methylamino)pyridines was necessary for the present C–H addition reaction catalyzed by a cationic iridium/chiral bisphosphine complex.
- This article is part of the themed collection: Functionalization of unreactive C-H bonds


 Please wait while we load your content...
Please wait while we load your content...