Abstract
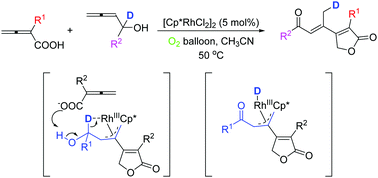
Herein we report a [Cp*RhCl2]2-catalyzed coupling cyclization of two different classes of allenes with 2,3-allenoic acids affording 2(5H)-furanone skeletons of products and 2,3-allenols forming a conjugated (E)-enal or enone functionality to the β-position of the 2(5H)-furanones. These products are important building blocks for the syntheses of potentially bioactive compounds. The reaction proceeded via the nucleometalation, insertion, and stereodefined 1,4-H delivery carried by rhodium.


 Please wait while we load your content...
Please wait while we load your content...