Fusing pyrene and ferrocene into a chiral, redox-active triangle†
Abstract
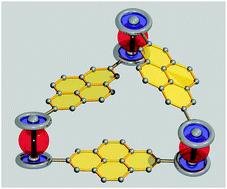
A macrocycle that integrates three ferrocene–pyrene dyads in a triangular C2-symmetric arrangement is synthesised as a racemate in a simple one-pot approach. Crystal structural analysis reveals two enantiomeric conformers that pack alternatingly via π–π stacking and interconvert dynamically in solution. Electrochemical investigations indicate weak electrostatic interactions between Fc groups upon oxidation to a mixed valence triangle.
- This article is part of the themed collection: Chemical Communications HOT articles


 Please wait while we load your content...
Please wait while we load your content...