Step-by-step dual stimuli-responsive nanoparticles for efficient bacterial biofilm eradication
Abstract
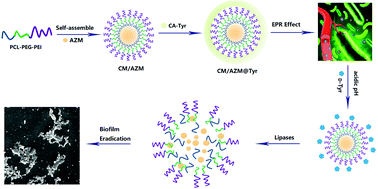
Biofilm-related bacterial infections are extremely resistant to antibiotics, mainly due to the impermeability of the intensive matrices, which allow the bacteria to survive antibiotic treatment. Herein, step-by-step dual stimuli-responsive azithromycin-loaded nanoparticles (CM/AZM@Tyr) was constructed for efficient biofilm eradication. CM/AZM@Tyr was prepared by the self-assembly of poly(ε-caprolactone)-polyethylene glycol-polyethylenimine (PCL-PEG-PEI) into cationic micelles and simultaneously encapsulated AZM into the hydrophobic core, which is further bound with cis-aconityl-D-tyrosine (CA-Tyr) through electrostatic interaction. Upon initial penetration, CM/AZM@Tyr could show step-by-step dual-response to the microenvironment of biofilms. Firstly, the CA-Tyr shell rapidly responded to the acidic microenvironment and released D-Tyr to disassemble the biofilm mass. Then, the exposed cationic CM/AZM micelles could bind firmly to the negatively-charged bacteria cell membrane. With the enzymolysis of the PCL core, the rapidly releasing AZM could kill the bacteria over the depth of biofilms. Massive accumulation was observed in the infected lungs of biofilms-associated lung infection mice after the i.v. injection of CM/Cy5.5@Tyr under the 3D mode of the in vivo Imaging System. Reduced bacterial burden and alleviated fibrosis in the infected lungs were also obtained after treatment with CM/AZM@Tyr mainly due to its intensive penetration in the biofilm and the orderly release of the biofilm dispersant and antimicrobial agents. In summary, this research developed an effective strategy for the treatment of blood-accessible biofilm-induced infections.

 Please wait while we load your content...
Please wait while we load your content...