Deep eutectic solvent decomposition-based microextraction for chromium determination in aqueous environments by atomic absorption spectrometry with electrothermal atomization†
Abstract
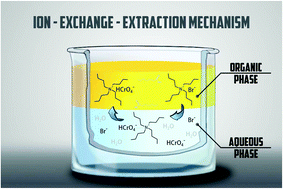
A sensitive, rapid, and simple procedure for the determination of traces of chromium species in natural and waste waters after microextraction using a quasi-hydrophobic deep eutectic solvent based on tetrabutylammonium bromide and hexanoic acid was developed for the first time. In the developed procedure, the deep eutectic solvent played the role of a source of dispersive agent, chelating agent, and extraction solvent. During mixing the aqueous phase with the quasi-hydrophobic deep eutectic solvent, dissolution and dissociation of tetrabutylammonium bromide took place. Tetrabutylammonium bromide acted as a dispersive agent for the hexanoic acid emulsion formation and as an agent for the formation of an ion-association complex with Cr(VI) in an aqueous phase followed by its extraction in hexanoic acid. The organic phase containing Cr(VI) complexes was analyzed by atomic absorption spectrometry with electrothermal atomization. The enrichment factor value was 53, the extraction recovery was 89%, and the limit of detection calculated from a blank test, based on 3σ, was 5.0 ng L−1. The values of intra-day RSD and inter-day RSD were 3.9% and 5.0%, respectively.
- This article is part of the themed collection: Analyst HOT Articles 2021


 Please wait while we load your content...
Please wait while we load your content...