Photocleavable core cross-linked polymeric micelles of polypept(o)ides and ruthenium(ii) complexes†
Abstract
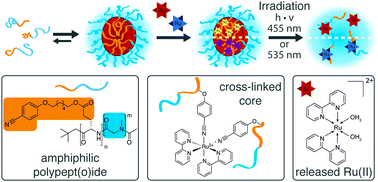
Core cross-linking of polymeric micelles has been demonstrated to contribute to enhanced stability that can improve the therapeutic efficacy. Photochemistry has the potential to provide spatial resolution and on-demand drug release. In this study, light-sensitive polypyridyl-ruthenium(II) complexes were combined with polypept(o)ides for photocleavable core cross-linked polymeric micelles. Block copolymers of polysarcosine-block-poly(glutamic acid) were synthesized by ring-opening N-carboxyanhydride polymerization and modified with aromatic nitrile-groups on the glutamic acid side chain. The modified copolymers self-assembled into micelles and were cross-linked by cis-diaquabis(2,2′-bipyridine)-ruthenium(II) ([Ru(bpy)2(H2O)2]2+) or cis-diaquabis(2,2′-biquinoline)-ruthenium(II) ([Ru(biq)2(H2O)2]2+). Depending on the flexibility and hydrophobicity of the nitrile linker, either small spherical structures (Dh 45 nm, PDI 0.11) or worm-like micelles were obtained. The cross-linking reaction did not affect the overall size distribution but induced a change in the metal-to-ligand charge transfer peak from 482 to 420 nm and 592 to 548 nm. The cross-linked micelles displayed colloidal stability after incubation with human blood plasma and during gel permeation chromatography in hexafluoroisopropanol. Light-induced cleavage of [Ru(bpy)2(H2O)2]2+ was accomplished within 300 s, while [Ru(biq)2(H2O)2]2+ could not be completely released. Analysis in HuH-7 cells revealed increased cytotoxicity via micellar delivery of [Ru(bpy)2(H2O)2]2+ but mostly irradiation damage for [Ru(biq)2(H2O)2]2+. Further evaluation in ovo confirmed stable circulation pointing towards the future development of quick-release complexes.


 Please wait while we load your content...
Please wait while we load your content...