In vitro and in vivo blood compatibility of concentrated polymer brushes†
Abstract
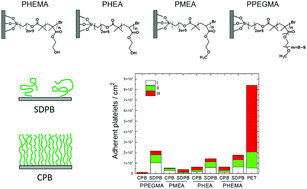
Concentrated polymer brushes (CPBs) and semi-dilute polymer brushes (SDPBs) of poly(2-hydroxyethyl methacrylate), poly(2-hydroxyethyl acrylate), poly[poly(ethylene glycol)methyl ether methacrylate] (PPEGMA) and poly(2-methoxyetyl acrylate) were prepared on silica particles and silicon wafers by surface-initiated atom transfer radical polymerization (SI-ATRP). In order to evaluate in vitro blood compatibility, plasma protein adsorption on the brushes was quantified with a BCA protein assay, and the adsorbed proteins on the brushes were identified using high-performance liquid chromatography coupled with tandem mass spectrometry (LC-MS/MS). All four CPBs displayed much less protein adsorption than their corresponding SDPBs. Interestingly, the number and type of identified proteins differed on the brushes. Platelet adhesion was then examined on the brushes, whereby CPBs suppressed platelet adhesion to a greater extent than the corresponding SDPBs, although platelet activation was observed on all surfaces. As a result, the CPBs of PPEGMA prevented platelet adhesion the most. After screening the polymers by in vitro evaluation, CPBs of PPEGMA were then grafted on a catheter by SI-ATRP. The catheter with the CPBs was implanted into the jugular vein of a rabbit. The in vivo assessment after three weeks of implantation confirmed that the CPBs caused little coagulation or inflammation, whereas the pristine catheter exhibited inflammation and encapsulation.


 Please wait while we load your content...
Please wait while we load your content...