Efficient chemical fixation of CO2 from direct air under environment-friendly co-catalyst and solvent-free ambient conditions†
Abstract
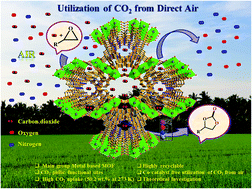
The capture and conversion of CO2 from direct air into value-added products under mild conditions represents a promising step towards environmental remediation and energy sustainability. Consequently, herein, we report the first example of a Mg(II)-based MOF exhibiting highly efficient fixation of CO2 from direct air into value-added cyclic carbonates under eco-friendly co-catalyst and solvent-free mild conditions. The bifunctional MOF catalyst was rationally constructed by utilizing an eco-friendly Lewis acidic metal ion, Mg(II), and a nitrogen-rich tripodal linker, TATAB. The MOF possesses a high BET surface area of 2606.13 m2 g−1 and highly polar 1D channels decorated with a high density of CO2-philic sites which promote a remarkably high CO2 uptake of 50.2 wt% at 273 K with a high heat of adsorption value of 55.13 kJ mol−1. The high CO2-affinity combined with the presence of a high density of nucleophilic and Lewis acidic sites conferred efficient catalytic properties to the Mg-MOF for chemical fixation of CO2 from direct air under environment-friendly mild conditions. The remarkable performance of the Mg-MOF for the fixation of CO2 from direct air was further supported by in-depth theoretical calculations. Moreover, the computational studies provided an insight into the mechanistic details of the catalytic process in the absence of any co-catalyst and solvent. Overall, this work represents a rare demonstration of carbon capture and utilization (CCU) from direct air under eco-friendly mild conditions.


 Please wait while we load your content...
Please wait while we load your content...