When water becomes an integral part of carbon – combining theory and experiment to understand the zeolite-like water adsorption properties of porous C2N materials†
Abstract
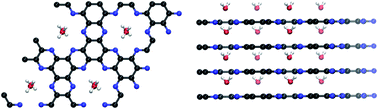
The interaction between water molecules and surfaces in porous systems is of huge importance in various fields including but not limited to catalysis, adsorption, and the storage or conversion of electrochemical energy. Hydrophilicity of usually non-polar carbon-based materials can be enhanced by the incorporation of heteroatoms like nitrogen but insights into the interaction mechanisms on a molecular level remain hardly achievable due to the lack of porous carbons with a defined and regular atomic construction and a suitable structure model for such substances. This makes theoretical calculations difficult. In order to illuminate the interactions between nitrogen-containing carbon surfaces and water molecules, water adsorption is studied in this work on a series of model materials with different pore structures and atomic construction. In particular, volumetric adsorption measurements and 1H solid-state NMR spectroscopic investigations in combination with theoretical calculations on water in nitrogen-rich C2N materials show a remarkably strong heat of adsorption significantly above 60 kJ mol−1, that is, far beyond what is typically considered physisorption. In such porous materials, water is becoming an integral part of the chemical architecture, and the term “zeocarbons” is therefore well deserved. From a general perspective, this study is intended to set a base for establishing the chemical principles of a new water chemistry confined in strongly interacting nanopores.


 Please wait while we load your content...
Please wait while we load your content...