Size-dependent hydrogen trapping in palladium nanoparticles†
Abstract
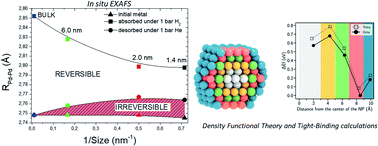
We report an experimental study, supported by a theoretical approach based on simulations, to explore the phenomenon of H trapping in small Pd nanoparticles. Hydrogen absorption/desorption of a series of Pd nanoparticles with different average sizes (6.0, 2.0 and 1.4 nm) is only partially reversible, as proven by pressure-composition-isotherms at 25 °C. The irreversible H amount is trapped into strong interstitial sites. In situ EXAFS was employed to highlight the local structural changes and the H trapping inside the volume of Pd nanoparticles. We evidence a double size-dependent effect of H trapping inside Pd nanoparticles: the smaller the Pd particle size, the larger the amount of trapped H and the higher the binding energy experienced by H atoms. For example, 26% of the initial H capacity is trapped in Pd nanoparticles with 2.0 and 1.4 nm average sizes and a treatment under vacuum above 150 °C is needed to fully desorb the trapped H. To get atomic-scale insights into the location of the trapping sites, we perform both density functional theory and tight-binding calculations. These simulations show that the strong H trapping sites are the octahedral interstitials located at the subsurface of Pd nanoparticles where high relaxations are possible. This finding might clarify the controversial role of “subsurface or bulk” hydrogen in Pd-based nano-catalysts in several hydrogenation reactions.


 Please wait while we load your content...
Please wait while we load your content...