Transient mechanical interactions between cells and viscoelastic extracellular matrix†
Abstract
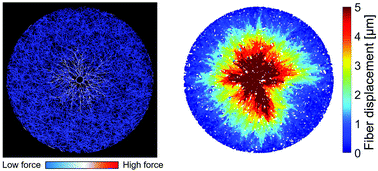
During various physiological processes, such as wound healing and cell migration, cells continuously interact mechanically with a surrounding extracellular matrix (ECM). Contractile forces generated by the actin cytoskeleton are transmitted to a surrounding ECM, resulting in structural remodeling of the ECM. To better understand how matrix remodeling takes place, a myriad of in vitro experiments and simulations have been performed during recent decades. However, physiological ECMs are viscoelastic, exhibiting stress relaxation or creep over time. The time-dependent nature of matrix remodeling induced by cells remains poorly understood. Here, we employed a discrete model to investigate how the viscoelastic nature of ECMs affects matrix remodeling and stress profiles. In particular, we used explicit transient cross-linkers with varied density and unbinding kinetics to capture viscoelasticity unlike most of the previous models. Using this model, we quantified the time evolution of generation, propagation, and relaxation of stresses induced by a contracting cell in an ECM. It was found that matrix connectivity, regulated by fiber concentration and cross-linking density, significantly affects the magnitude and propagation of stress and subsequent matrix remodeling, as characterized by fiber displacements and local net deformation. In addition, we demonstrated how the base rate and force sensitivity of cross-linker unbinding regulate stress profiles and matrix remodeling. We verified simulation results using in vitro experiments performed with fibroblasts encapsulated in a three-dimensional collagen matrix. Our study provides key insights into the dynamics of physiologically relevant mechanical interactions between cells and a viscoelastic ECM.
- This article is part of the themed collection: Force transmission by nonlinear biomaterials


 Please wait while we load your content...
Please wait while we load your content...