Fine-tuning the water oxidation performance of hierarchical Co3O4 nanostructures prepared from different cobalt precursors†
Abstract
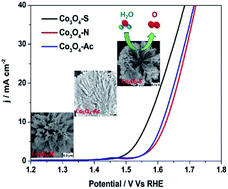
The design of efficient and low-cost catalysts for water oxidation with superior electrochemical performance is of great importance. Herein, we report on the fine-tuning of water oxidation performance using solvothermal fabricated hierarchical nanostructured Co3O4 catalysts obtained from three different cobalt precursors. The prepared hierarchical Co3O4 nanostructured catalysts were intensively analyzed for their textural properties and surface chemistry, such as shape, size, phase and surface area. The water oxidation performance of all three catalysts through the oxygen evolution reaction (OER) has been investigated. In alkaline 1 M KOH aqueous solution, the ultrathin hierarchical Co3O4–S nanosheet array catalyst exhibits significantly higher activity during water oxidation, with a low overpotential of 330 mV vs. RHE at a benchmarking current density of 10 mA cm−2 for OER in comparison with Co3O4–Ac and Co3O4–N, which outperforms a commercial RuO2 electrocatalyst, and it remains stable for many hours. Benefiting from its unique ultrathin architecture, hierarchical and nanostructured Co3O4–S is endowed with a large number of active sites and a rapid charge transfer process, resulting in better water oxidation performance.


 Please wait while we load your content...
Please wait while we load your content...