CsPbBr3–CdS heterostructure: stabilizing perovskite nanocrystals for photocatalysis†
Abstract
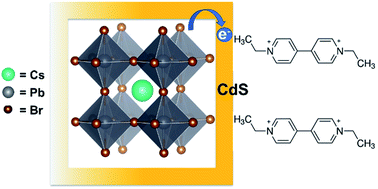
The instability of cesium lead bromide (CsPbBr3) nanocrystals (NCs) in polar solvents has hampered their use in photocatalysis. We have now succeeded in synthesizing CsPbBr3–CdS heterostructures with improved stability and photocatalytic performance. While the CdS deposition provides solvent stability, the parent CsPbBr3 in the heterostructure harvests photons to generate charge carriers. This heterostructure exhibits longer emission lifetime (τave = 47 ns) than pristine CsPbBr3 (τave = 7 ns), indicating passivation of surface defects. We employed ethyl viologen (EV2+) as a probe molecule to elucidate excited state interactions and interfacial electron transfer of CsPbBr3–CdS NCs in toluene/ethanol mixed solvent. The electron transfer rate constant as obtained from transient absorption spectroscopy was 9.5 × 1010 s−1 and the quantum efficiency of ethyl viologen reduction (ΦEV+˙) was found to be 8.4% under visible light excitation. The Fermi level equilibration between CsPbBr3–CdS and EV2+/EV+˙ redox couple has allowed us to estimate the apparent conduction band energy of the heterostructure as −0.365 V vs. NHE. The insights into effective utilization of perovskite nanocrystals built around a quasi-type II heterostructures pave the way towards effective utilization in photocatalytic reduction and oxidation processes.


 Please wait while we load your content...
Please wait while we load your content...