Total synthesis and mechanism of action of the antibiotic armeniaspirol A†
Abstract
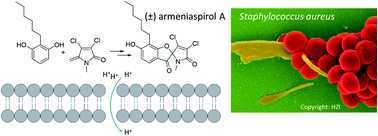
Emerging antimicrobial resistance urges the discovery of antibiotics with unexplored, resistance-breaking mechanisms. Armeniaspirols represent a novel class of antibiotics with a unique spiro[4.4]non-8-ene scaffold and potent activities against Gram-positive pathogens. We report a concise total synthesis of (±) armeniaspirol A in six steps with a yield of 20.3% that includes the formation of the spirocycle through a copper-catalyzed radical cross-coupling reaction. In mechanistic biological experiments, armeniaspirol A exerted potent membrane depolarization, accounting for the pH-dependent antibiotic activity. Armeniaspirol A also disrupted the membrane potential and decreased oxygen consumption in mitochondria. In planar lipid bilayers and in unilamellar vesicles, armeniaspirol A transported protons across membranes in a protein-independent manner, demonstrating that armeniaspirol A acted as a protonophore. We provide evidence that this mechanism might account for the antibiotic activity of multiple chloropyrrole-containing natural products isolated from various origins that share a 4-acylphenol moiety coupled to chloropyrrole as a joint pharmacophore. We additionally describe an efflux-mediated mechanism of resistance against armeniaspirols.


 Please wait while we load your content...
Please wait while we load your content...