Photoactive electron donor–acceptor complex platform for Ni-mediated C(sp3)–C(sp2) bond formation†
Abstract
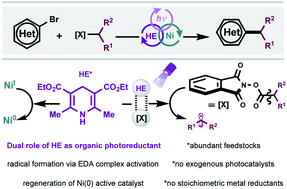
A dual photochemical/nickel-mediated decarboxylative strategy for the assembly of C(sp3)–C(sp2) linkages is disclosed. Under light irradiation at 390 nm, commercially available and inexpensive Hantzsch ester (HE) functions as a potent organic photoreductant to deliver catalytically active Ni(0) species through single-electron transfer (SET) manifolds. As part of its dual role, the Hantzsch ester effects a decarboxylative-based radical generation through electron donor–acceptor (EDA) complex activation. This homogeneous, net-reductive platform bypasses the need for exogenous photocatalysts, stoichiometric metal reductants, and additives. Under this cross-electrophile paradigm, the coupling of diverse C(sp3)-centered radical architectures (including primary, secondary, stabilized benzylic, α-oxy, and α-amino systems) with (hetero)aryl bromides has been accomplished. The protocol proceeds under mild reaction conditions in the presence of sensitive functional groups and pharmaceutically relevant cores.
- This article is part of the themed collections: Most popular 2021 organic chemistry articles, 2021 and 2021 Chemical Science HOT Article Collection


 Please wait while we load your content...
Please wait while we load your content...