CHIP-mediated hyperubiquitylation of tau promotes its self-assembly into the insoluble tau filaments†
Abstract
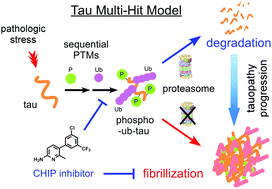
The tau protein is a highly soluble and natively unfolded protein. Under pathological conditions, tau undergoes multiple post-translational modifications (PTMs) and conformational changes to form insoluble filaments, which are the proteinaceous signatures of tauopathies. To dissect the crosstalk among tau PTMs during the aggregation process, we phosphorylated and ubiquitylated recombinant tau in vitro using GSK3β and CHIP, respectively. The resulting phospho–ub-tau contained conventional polyubiquitin chains with lysine 48 linkages, sufficient for proteasomal degradation, whereas unphosphorylated ub-tau species retained only one–three ubiquitin moieties. Mass-spectrometric analysis of in vitro reconstituted phospho–ub-tau revealed seven additional ubiquitylation sites, some of which are known to stabilize tau protofilament stacking in the human brain with tauopathy. When the ubiquitylation reaction was prolonged, phospho–ub-tau transformed into insoluble hyperubiquitylated tau species featuring fibrillar morphology and in vitro seeding activity. We developed a small-molecule inhibitor of CHIP through biophysical screening; this effectively suppressed tau ubiquitylation in vitro and delayed its aggregation in cultured cells including primary cultured neurons. Our biochemical findings point to a “multiple-hit model,” where sequential events of tau phosphorylation and hyperubiquitylation function as a key driver of the fibrillization process, thus indicating that targeting tau ubiquitylation may be an effective strategy to alleviate the course of tauopathies.


 Please wait while we load your content...
Please wait while we load your content...