Trapped interfacial redox introduces reversibility in the oxygen reduction reaction in a non-aqueous Ca2+ electrolyte†
Abstract
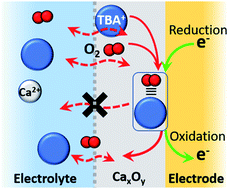
Electrochemical investigations of the oxygen reduction reaction (ORR) and oxygen evolution reaction (OER) have been conducted in a Ca2+-containing dimethyl sulfoxide electrolyte. While the ORR appears irreversible, the introduction of a tetrabutylammonium perchlorate (TBAClO4) co-salt in excess concentrations results in the gradual appearance of a quasi-reversible OER process. Combining the results of systematic cyclic voltammetry investigations, the degree of reversibility depends on the ion pair competition between Ca2+ and TBA+ cations to interact with generated superoxide (O2−). When TBA+ is in larger concentrations, and large reductive overpotentials are applied, a quasi-reversible OER peak emerges with repeated cycling (characteristic of formulations without Ca2+ cations). In situ Raman microscopy and rotating ring-disc electrode (RRDE) experiments revealed more about the nature of species formed at the electrode surface and indicated the progressive evolution of a charge storage mechanism based upon trapped interfacial redox. The first electrochemical step involves generation of O2−, followed primarily by partial passivation of the surface by CaxOy product formation (the dominant initial reaction). Once this product matrix develops, the subsequent formation of TBA+--O2− is contained within the CaxOy product interlayer at the electrode surface and, consequently, undergoes a facile oxidation reaction to regenerate O2.
- This article is part of the themed collections: Materials for energy storage and conversion: Chemical Science symposium collection and Editor’s Choice: Zaiping Guo


 Please wait while we load your content...
Please wait while we load your content...