Discovery and biosynthesis of guanipiperazine from a NRPS-like pathway†
Abstract
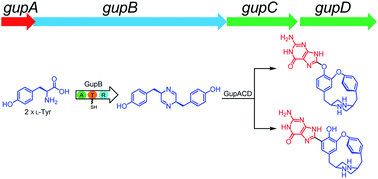
Nonribosomal peptide synthetases (NRPSs) are modular enzymes that use a thiotemplate mechanism to assemble the peptide backbones of structurally diverse and biologically active natural products in bacteria and fungi. Unlike these canonical multi-modular NRPSs, single-module NRPS-like enzymes, which lack the key condensation (C) domain, are rare in bacteria, and have been largely unexplored to date. Here, we report the discovery of a gene cluster (gup) encoding a NRPS-like megasynthetase through genome mining. Heterologous expression of the gup cluster led to the production of two unprecedented alkaloids, guanipiperazines A and B. The NRPS-like enzyme activates two L-tyrosine molecules, reduces them to the corresponding amino aldehydes, and forms an unstable imine product. The subsequent enzymatic reduction affords piperazine, which can be morphed by a P450 monooxygenase into a highly strained compound through C–O bond formation. Further intermolecular oxidative coupling forming the C–C or C–O bond is catalyzed by another P450 enzyme. This work reveals the huge potential of NRPS-like biosynthetic gene clusters in the discovery of novel natural products.


 Please wait while we load your content...
Please wait while we load your content...