Distinct electronic structures and bonding interactions in inverse-sandwich samarium and ytterbium biphenyl complexes†
Abstract
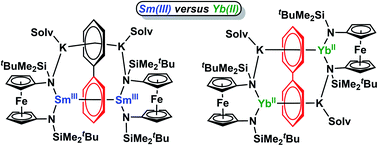
Inverse-sandwich samarium and ytterbium biphenyl complexes were synthesized by the reduction of their trivalent halide precursors with potassium graphite in the presence of biphenyl. While the samarium complex had a similar structure as previously reported rare earth metal biphenyl complexes, with the two samarium ions bound to the same phenyl ring, the ytterbium counterpart adopted a different structure, with the two ytterbium ions bound to different phenyl rings. Upon the addition of crown ether to encapsulate the potassium ions, the inverse-sandwich samarium biphenyl structure remained intact; however, the ytterbium biphenyl structure fell apart with the concomitant formation of a divalent ytterbium crown ether complex and potassium biphenylide. Spectroscopic and computational studies were performed to gain insight into the electronic structures and bonding interactions of these samarium and ytterbium biphenyl complexes. While the ytterbium ions were found to be divalent with a 4f14 electron configuration and form a primarily ionic bonding interaction with biphenyl dianion, the samarium ions were in the trivalent state with a 4f5 electron configuration and mainly utilized the 5d orbitals to form a δ-type bonding interaction with the π* orbitals of the biphenyl tetraanion, showing covalent character.


 Please wait while we load your content...
Please wait while we load your content...