Dihydronicotinamide riboside: synthesis from nicotinamide riboside chloride, purification and stability studies†
Abstract
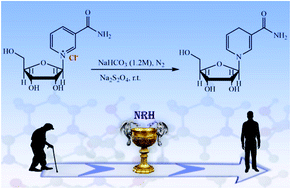
In the present work, we describe an efficient method for scalable synthesis and purification of 1,4-dihydronicotinamide riboside (NRH) from commercially available nicotinamide riboside chloride (NRCl) and in the presence of sodium dithionate as a reducing agent. NRH is industrially relevant as the most effective, synthetic NAD+ precursor. We demonstrated that solid phase synthesis cannot be used for the reduction of NRCl to NRH in high yield, whereas a reduction reaction in water at room temperature under anaerobic conditions is shown to be very effective, reaching a 55% isolation yield. For the first time, by using common column chromatography, we were able to highly purify this sensitive bio-compound with good yield. A series of identifications and analyses including HPLC, NMR, LC-MS, FTIR, and UV-vis spectroscopy were performed on the purified sample, confirming the structure of NRH as well as its purity to be 96%. Thermal analysis of NRH showed higher thermal stability compared to NRCl, and with two major weight losses, one at 218 °C and another at 805 °C. We also investigated the long term stability effects of temperature, pH, light, and oxygen (as air) on the NRH in aqueous solutions. Our results show that NRH can be oxidized in the presence of oxygen, and it hydrolyzed quickly in acidic conditions. It was also found that the degradation rate is lower under a N2 atmosphere, at lower temperatures, and under basic pH conditions.


 Please wait while we load your content...
Please wait while we load your content...