Blackening of titanium dioxide nanoparticles by atomic hydrogen and the effect of coexistence of water on the blackening
Abstract
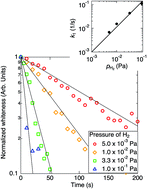
A fast blackening process of titanium dioxide nanoparticles by exposing to atomic hydrogen was studied by estimating the color of the nanoparticles. The whiteness of TiO2 decreased exponentially with time, which suggests a first-order reaction between atomic H and surface oxygen, whose rate constant is proportional to the ambient pressure of H2. The rate constant increases as the temperature of nanoparticles at exposing to atomic hydrogen. The structure and size of nanoparticles were estimated by the X-ray diffraction (XRD), which shows that a part of anatase transferred to rutile and the crystal sizes of both anatase and rutile increased by hydrogenation above 600 K. The blackening of TiO2 halfway stopped under the condition of the similar partial pressure of water with hydrogen. This suggests the presence of reverse reaction between H2O and oxygen vacancy, whose reaction rate constant is proportional to the partial pressure of H2O.


 Please wait while we load your content...
Please wait while we load your content...