Metastable Zr/Hf-MOFs: the hexagonal family of EHU-30 and their water-sorption induced structural transformation†
Abstract
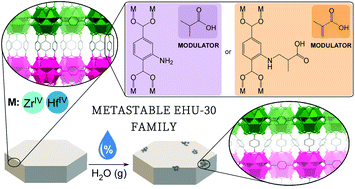
UiO-66, reported in 2008, inspired the ongoing chemistry of group 4 metal–organic frameworks and it is still one of the most studied MOFs due to its exceptional stability. The tightness of their building blocks and bond strength make it tough to access new topologies, unless the building blocks are changed or defect engineering is employed, which in turn lead to different nominal stoichiometries. However, well selected structure directing agents and harsh synthesis conditions allowed the isolation of the first polymorph of UiO-66, named EHU-30. Herein, we provide evidence for the generalization of the synthetic approach with four new EHU-30 isoreticular compounds, based on amino-functionalized linkers and zirconium and hafnium as metal centres: EHU-30-NH2(M) and EHU-30-NHR(M) (M: Zr, Hf; R: 2-carboxypropyl). The crystal structure analysis reveals that their framework is more porous than that of their polymorphic counterpart. Furthermore, due to the current relevance of MOFs in greenhouse gas capture and in water harvesting methodologies, we have explored CO2 and water vapour adsorption on the new polymorphic phases, by means of experimental and computational resources, and compared to the parent EHU-30. The amino functionalization makes the EHU-30 series more prone to CO2 and water vapour adsorption. Besides, the water adsorption isotherms show an interesting crossing sensitive to temperature, sorption cycles and functionalization of the linker, which has been attributed to a localized structural transformation from EHU-30 to UiO-66. Finally, time-of-flight (TOF) powder neutron diffraction experiments were also conducted to locate the preferred adsorption sites for water in functionalized and parent EHU-30 structures.


 Please wait while we load your content...
Please wait while we load your content...