Heterogeneity induced dual luminescence properties of AgInS2 and AgInS2–ZnS alloyed nanocrystals†
Abstract
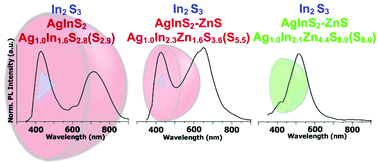
We have elaborated a new preparation method of ternary AgInS2 and alloyed quaternary AgInS2–ZnS nanocrystals which consisted of two consecutive injections of sulfur (S dissolved in oleylamine, OLA) and silver (AgNO3 or Ag2CO3 dissolved in dichlorobenzene, DCB) precursors into a mixture of indium(III) chloride, zinc stearate and 1-dodecanethiol (DDT) dissolved in 1-octadecene (ODE). Under these conditions, nucleation of cubic In2S3 seeds took place followed by the growth of orthorhombic AgInS2 or alloyed AgInS2–ZnS cubic phases to yield heterodimer type nanocrystals. In both types of nanocrystals clearly separated photoluminescence peaks could be observed, confirming their heterogenic nature. The first one at 430 nm originated from the luminescence of the In2S3 phase. The second one was ascribed to the presence of a ternary Ag–In–S or quaternary Ag–In–Zn–S phase and its position, within the spectroscopic range from 515 nm to 710 nm, strongly dependent on the nanocrystal composition. The registered two-dimensional excitation–emission topographical maps clearly indicated that the observed emissions in two different spectral regions were related to the excitation in the same spectral range (300–400 nm); however their photoluminescence mechanisms were distinctly different. The photoluminescence lifetime of ∼3 ns measured for the emission at shorter wavelengths was typical of the simple mechanism of exciton radiative recombination in the In2S3 phase. The significantly longer lifetime of the longer wavelength emission (∼26 μs) seemed to clearly indicate that in this case the photoluminescence mechanism was more complex, involving exciton trap states whose positions depended on the composition of the ternary (or quaternary) phase.


 Please wait while we load your content...
Please wait while we load your content...