Acid-triggering of light-induced charge-separation in hybrid organic/inorganic molecular photoactive dyads for harnessing solar energy†
Abstract
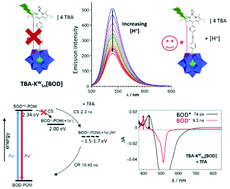
H+ modulated charge-transfer in photoexcited covalent polyoxometalate-bodipy conjugates is described. The hybrid organic/inorganic molecular photoactive dyads are based on Keggin-type polyoxometalates (POMs, where KM = [PM11O39] and M = Mo or W) covalently grafted via an organotin linker to a bodipy (BOD) photosensitizer. The relative potentials of the photosensitizer and POM are aligned such that light-induced electron transfer from BOD to POM is permitted for the polyoxomolybdate KMoSn[BOD] but not effective for the polyoxotungstate analogue KWSn[BOD]. In both cases, the addition of acid shifts the redox potential of the POM only, to increase the driving force for electron transfer. This leads to charge-separation being switched on for KWSn[BOD] in the presence of acid. The addition of acid to KMoSn[BOD] accelerates charge-separation by an order of magnitude (from 2 ns to 200 ps) and is accompanied by a deceleration of charge recombination, leading to a charge-separated state lifetime of up to 1.3 μs. This behaviour is consistent with proton coupled electron transfer, which has previously been observed electrochemically for POMs, but this study shows, for the first time, the impact of protonation on photoinduced electron transfer.


 Please wait while we load your content...
Please wait while we load your content...