Chemical conjugation of nucleic acid aptamers and synthetic polymers
Abstract
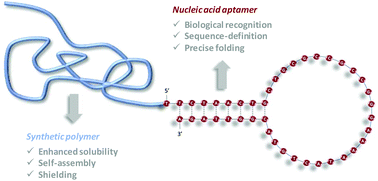
Nucleic acid aptamers are chemically-synthesized single-stranded oligonucleotides that fold into specific sequence-dependent configurations. Due to their exceptional recognition properties towards a variety of biological targets, they find applications in many areas of bioscience such as affinity chromatography, drug delivery, biosensors, diagnostics, stem cell research and regenerative medicine. In recent years, aptamers conjugation to synthetic polymers has gained increasing attention, as an effective strategy to control biochemical parameters, uptake mechanisms and pharmacokinetic properties. In this review, we summarize and critically discuss the different approaches that have been used so far for the preparation of aptamer–polymer conjugates. Most of the reported work has been achieved by reacting an aptamer with a functional polymer in solution. Different ligation chemistries such as active ester amidation, thio-Michael addition, direct thiol–disulfide exchange and azide–alkyne Huisgen cycloaddition have been used for that purpose. Alternatively, polymer conjugation can be performed on solid-supports. After chemical synthesis, resin bound aptamers are reacted with a macromolecular reagent or extended by phosphoramidite polymer chemistry. Furthermore, some non-covalent strategies, such as polyelectrolyte complexation, have been reported for the preparation of aptamer–polymer conjugates. The pro and cons of all these strategies are discussed herein.


 Please wait while we load your content...
Please wait while we load your content...