Sequence-regulated vinyl polymers via iterative atom transfer radical additions and acyclic diene metathesis polymerization†
Abstract
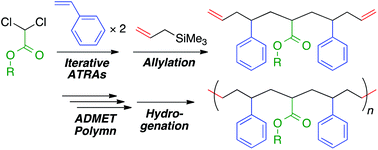
A new strategy for the synthesis of sequence-regulated vinyl polymers based on a combination of iterative atom transfer radical additions (ATRAs) of vinyl monomers and acyclic diene metathesis (ADMET) polymerization was developed. A molecularly defined vinyl monomer sequence at the single monomer unit level was constructed in telechelic diene sequence-regulated oligomers by iterative metal-catalyzed ATRAs of vinyl monomers to dichloride followed by allylation of both carbon–chloride terminals. ADMET polymerization of the obtained telechelic diene oligomer with a symmetrical monomer sequence resulted in polymers with an embedded vinyl monomer sequence that was repeatedly connected by an olefin formed by olefin metathesis polymerization. The subsequent hydrogenation of the main-chain olefins led to polymers that mimicked sequence-regulated vinyl polymer structures consisting of saturated C–C main chains. In particular, this study enabled the synthesis of sequence-regulated vinyl polymer mimics that have perfect and periodic styrene–acrylate–styrene–ethylene–methylene sequences, no statistical distributions of the comonomer compositions and sequences in polymer chains, relatively high molecular weights, and only carbon–carbon single bonds in the main chain. The thermal properties of the sequence-regulated vinyl polymer mimics were evaluated and compared with those of hypothetical random copolymers of styrene, acrylate, and ethylene units with the same comonomer contents.
- This article is part of the themed collections: Molecularly Defined Polymers: Synthesis and Function and Polymer Chemistry Most Popular 2021


 Please wait while we load your content...
Please wait while we load your content...