Structural evolution of iron forming iron oxide in a deep eutectic-solvothermal reaction†
Abstract
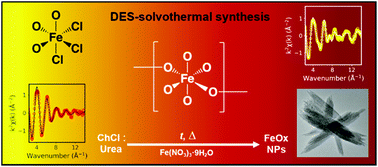
Deep eutectic solvents (DES) and their hydrated mixtures are used for solvothermal routes towards greener functional nanomaterials. Here we present the first static structural and in situ studies of the formation of iron oxide (hematite) nanoparticles in a DES of choline chloride : urea where xurea = 0.67 (aka. reline) as an exemplar solvothermal reaction, and observe the effects of water on the reaction. The initial speciation of Fe3+ in DES solutions was measured using extended X-ray absorption fine structure (EXAFS), while the atomistic structure of the mixture was resolved from neutron and X-ray diffraction and empirical potential structure refinement (EPSR) modelling. The reaction was monitored using in situ small-angle neutron scattering (SANS), to determine mesoscale changes, and EXAFS, to determine local rearrangements of order around iron ions. It is shown that iron salts form an octahedral [Fe(L)3(Cl)3] complex where (L) represents various O-containing ligands. Solubilised Fe3+ induced subtle structural rearrangements in the DES due to abstraction of chloride into complexes and distortion of H-bonding around complexes. EXAFS suggests the complex forms [–O–Fe–O–] oligomers by reaction with the products of thermal hydrolysis of urea, and is thus pseudo-zero-order in iron. In the hydrated DES, the reaction, nucleation and growth proceeds rapidly, whereas in the pure DES, the reaction initially proceeds quickly, but suddenly slows after 5000 s. In situ SANS and static small-angle X-ray scattering (SAXS) experiments reveal that nanoparticles spontaneously nucleate after 5000 s of reaction time in the pure DES before slow growth. Contrast effects observed in SANS measurements suggest that hydrated DES preferentially form 1D particle morphologies because of choline selectively capping surface crystal facets to direct growth along certain axes, whereas capping is restricted by the solvent structure in the pure DES.


 Please wait while we load your content...
Please wait while we load your content...