Hybrid multimodal contrast agent for multiscale in vivo investigation of neuroinflammation†
Abstract
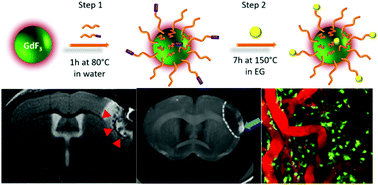
Neuroinflammation is a process common to several brain pathologies. Despites its medical relevance, it still remains poorly understood; there is therefore a need to develop new in vivo preclinical imaging strategies to monitor inflammatory processes longitudinally. We here present the development of a hybrid imaging nanoprobe named NP3, that was specifically designed to get internalized by phagocytic cells and imaged in vivo with MRI and bi-photon microscopy. NP3 is composed of a 16 nm core of gadolinium fluoride (GdF3), coated with bisphosphonate polyethylene glycol (PEG) and functionalized with a Lemke-type fluorophore. It has a hydrodynamic diameter of 28 ± 8 nm and a zeta potential of −42 ± 6 mV. The MR relaxivity ratio at 7 T is r1/r2 = 20; therefore, NP3 is well suited as a T2/T2* contrast agent. In vitro cytotoxicity assessments performed on four human cell lines revealed no toxic effects of NP3. In addition, NP3 is internalized by macrophages in vitro without inducing inflammation or cytotoxicity. In vivo, uptake of NP3 has been observed in the spleen and the liver. NP3 has a prolonged vascular remanence, which is an advantage for macrophage uptake in vivo. The proof-of-concept that NP3 may be used as a contrast agent targeting phagocytic cells is provided in an animal model of ischemic stroke in transgenic CX3CR1-GFP/+ mice using three complementary imaging modalities: MRI, intravital two-photon microscopy and phase contrast imaging with synchrotron X-rays. In summary, NP3 is a promising preclinical tool for the multiscale and multimodal investigation of neuroinflammation.


 Please wait while we load your content...
Please wait while we load your content...