Nuclearity growth of new PdII complexes induced by the electronic effect of selenium-containing ligands†
Abstract
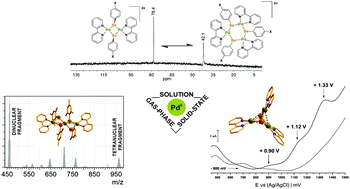
Five new PdII complexes are reported and have been obtained by the reaction of monomeric [Pd(bipy)(py)2]2+, which serves as a builing block, and substituted (ArSe)2 or Pd(SeAr)2. Complexes [Pd2(μ-SeAr)2(bipy)2](PF6)2, Ar = Ph (1), p-ClC6H4 (2), p-FC6H4 (3), [Pd3(μ3-Se)2(bipy)3](PF6)2·5THF (4) and [Pd3(μ-SeMes)4(bipy)2](PF6)2·2MeCN (5) showed that the nuclearity of these structures is affected by electron donating groups (EDGs) and electron withdrawing groups (EWGs) from the selenium-containing ligands, where changing the stoichiometry ratio of the reactants does not affect the number of palladium centers. The distinct behaviors of 1–5 in the solid state, solution phase and gas phase were investigated by single-crystal XRD, NMR and ESI-TOF-MS. In solution, the dinuclear 1–3 structures exist in equilibrium with trinuclear species. In the gas phase, trinuclear and tetranuclear aggregates of 1–3 are detected, as well as dinuclear and tetranuclear aggregates for 5, while compound 4 maintains the same nuclearity. The solid-state electrochemical behavior was studied, and all complexes were electrochemically active, exhibiting irreversible electrochemical processes, where selenium is indicated to be the active species which oxidizes under such conditions. The complexes are potential candidates to act as semiconductor sensitizers for photocatalytic hydrogen evolution, as they absorb in the visible region with optical band gap energies in the range of 2.6 eV.


 Please wait while we load your content...
Please wait while we load your content...