Osmapentalyne and osmapentalene complexes containing boron monofluoride ligands: structure, bonding and adaptive aromaticity†
Abstract
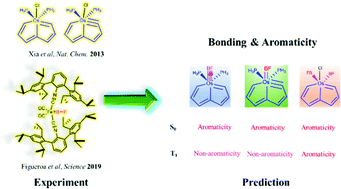
Osmapentalyne and osmapentalene complexes, now termed as carbolong species, have attracted considerable attention due to their novel structures, reactivities, and chelating properties as well as Möbius and adaptive aromaticity. On the other hand, boron monofluoride (BF), a 10-electron diatomic molecule isoelectronic to carbon monoxide (CO), is unstable below 1800 °C in the gas phase, and preparation of its metal complex is particularly challenging. Although the iron complex featuring a terminal BF ligand was synthesized recently, the structure, bonding and aromaticity of metallapentalyne and metallapentalene complexes containing BF ligands remain unclear. Herein, we carry out density functional theory (DFT) calculations to address this issue. The Os–B bond could be considered as a single, double or triple bond depending on the ligands attached to the metal center according to principal interacting orbital (PIO) analysis. The similarity on aromaticity is revealed between CO complexes and BF ones. All these species display aromaticity in the S0 state. For the T1 state, they could be aromatic, nonaromatic or antiaromatic depending on the excitation mode. All these findings could be useful to develop the concept of adaptive aromaticity as well as BF chemistry.


 Please wait while we load your content...
Please wait while we load your content...